Spring 2023 - Vol. 18, No. 1
SCIENTIFIC REPORT
Prevalence of Asymptomatic and
Symptomatic SARS-CoV-2 Infection
in a Universally Screened Obstetric Population
Loeven Bachilas Vernon
Michael R. Loeven, MD, MBA
Associate Director
Holly E. Bachilas, MD
Family Physician
LG Health Family Medicine Residency Program
Kellie Bresz, MS
Statistician
Tawnya Vernon, MPH
Former Project Manager
LG Health Research Institute
OBJECTIVE
The objective of this descriptive study is to determine the rate of symptomatic and asymptomatic carriers of SARS-CoV-2 in a universally screened population of patients admitted to labor and delivery at a suburban community hospital.
BACKGROUND
Early in the COVID-19 pandemic, accurate identification of potentially contagious individuals was recognized as important for adequate protection of hospital staff and appropriate management of patients. This was particularly true in the obstetric population due to a high frequency of contact with health care providers and the concern that the second stage of labor could be an aerosolizing event.1 Additionally, there was concern that the relatively younger population of laboring mothers would contain a significant number of asymptomatic carriers of the virus with associated potential for spread. It is now thought that at least one-third of SARS-CoV-2 infections in the general population are asymptomatic2 and that over half of all infections come through asymptomatic transmission, including presymptomatic and truly asymptomatic cases.3
Studies of obstetric patients admitted to New York City hospitals in March and April 2020 revealed a rate of asymptomatic positive patients as high as 14% to 33% of the presenting population.4-6 This finding led to the development of universal testing policies in many obstetric wards throughout the country. It was unclear, however, if these rates could be generalized to areas with comparatively lower prevalence of SARS-CoV-2 infection. Studies in obstetric patients in southern Connecticut and Boston in the spring of 2020 revealed a prevalence of asymptomatic positives of 2.9% and 1.5%, respectively.7,8
Penn Medicine Lancaster General Health Women & Babies Hospital began universal SARS-CoV-2 testing for obstetric patients admitted for delivery on April 22, 2020. At that time, Lancaster County had seen a relatively lower prevalence of SARS-CoV-2 infection compared to New York City.9 Prevalence waxed and waned throughout 2020 and peaked during the third wave of the disease in late 2020, though it remained lower than the peak prevalence rates at major epicenters (see Fig. 1).
 Fig. 1. New COVID cases in New York County, New York vs. Lancaster County, Pennsylvania from May 2020 through April 2021.
Fig. 1. New COVID cases in New York County, New York vs. Lancaster County, Pennsylvania from May 2020 through April 2021.
Graph by the LG Health Research Institute including data from Dong, Du, and Gardner, 2020.10
Region-specific data on asymptomatic carriers, particularly in lower prevalence areas such as Lancaster County, would be useful for anticipatory planning and conservation of tests and personal protective equipment (PPE). This is particularly true since the advent of vaccination and fluctuating prevalence of SARS-CoV-2 in the population at large.
METHODS
Study Design
A retrospective review of health records of all patients admitted for delivery to Women & Babies Hospital from April 22, 2020 through May 1, 2021 was performed. All subjects who were pregnant and presented at our facility for delivery during the study time period were included in the study. The start date denotes the time that universal screening was initiated for all labor and delivery admissions, including planned Cesarean delivery. Antepartum or other non-laboring admissions were excluded. A total of 4,221 subjects were enrolled from this single site, with 3,979 having a SARS-CoV-2 test within five days before delivery. Patients with SARS-CoV-2 tests completed after the date of admission were not included.
The electronic medical record for each patient that tested positive for SARS-CoV-2 was reviewed for documentation of the presence or absence of symptoms at the time of presentation. The documentation of any one symptom (including but not limited to fever, cough, shortness of breath, congestion, sore throat, and fatigue) was counted as a symptomatic infection. Vital signs were not directly used to determine if a patient was symptomatic or asymptomatic.
Patients were excluded from the study if they had a prior positive test 14 or more days before admission and therefore were no longer candidates for testing based on current infection-prevention protocols.
This study, reviewed and approved by the institutional review board, was granted a waiver of informed consent.
SARS-CoV-2 Testing Methods
All SARS-CoV-2 testing was performed by collecting nasopharyngeal swabs. Swabs were tested by the Cepheid GeneXpert testing system, which uses real-time reverse transcriptase polymerase chain reaction (RT-PCR) to detect RNA fragments of SARS-CoV-2. The test does not differentiate between potentially infectious and noninfectious genetic fragments. This test previously demonstrated a sensitivity of 100% and specificity of 80%.11
The rate of asymptomatic positive SARS-CoV-2 among laboring mothers was analyzed, and the rate of positive SARS-CoV-2 tests among laboring mothers was compared to the overall positive rate in the Lancaster General Health patient population, which includes non-obstetric patients in the same health network.
RESULTS
Demographic information on study subjects is summarized in Table 1. Body mass index (BMI) was the only demographic factor noted to be associated with a statistically significant increase in SARS-CoV-2 test positivity (p = 0.038).
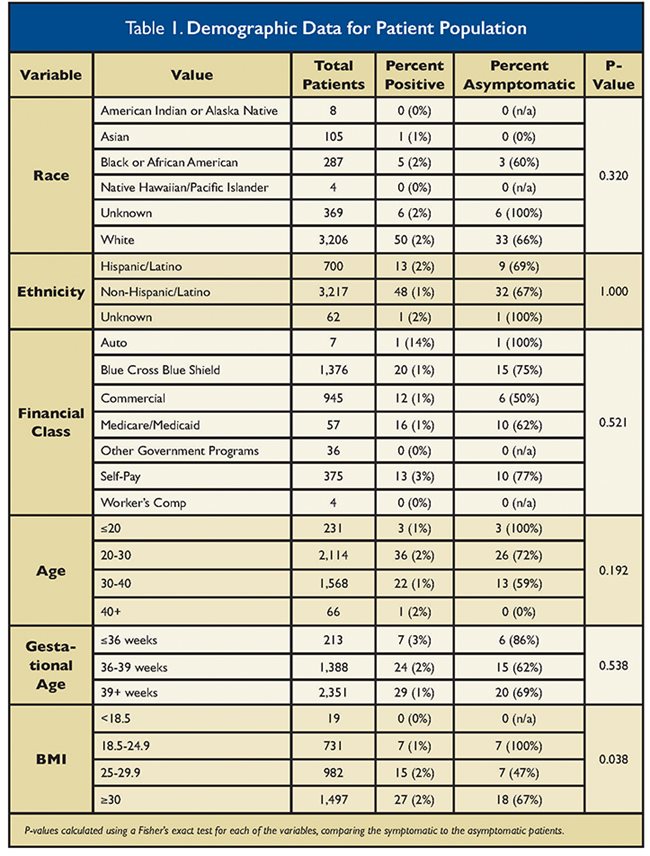
There were 62 positive tests in the cohort for a test positivity rate of 1.6% (62/3,979). Of those who tested positive, 68% (42/62) were determined to be asymptomatic. Of the symptomatic positive patients, 19 out of 20 had mild disease, with the most frequently reported symptom being mild nasal congestion. One patient had more severe symptoms, which required supplemental oxygen.
Fig. 2 summarizes the percent positivity and percent asymptomatic over the study period. The percent positive rate of tests among laboring mothers trended similarly over time while being lower than the overall percent positive rate of tests in the LG Health patient population. During times when the overall prevalence was high (particularly from October 2020 to February 2021), there were higher numbers of women presenting in labor who tested positive for SARS-CoV-2 and a higher percentage who were asymptomatic.
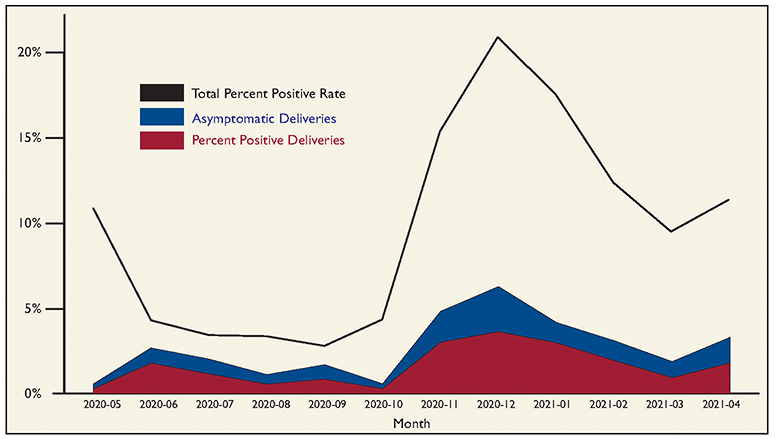
Fig. 2. Percent positive rate of SAR-CoV-2 among mothers who delivered (blue); asymptomatic rate (red) included within positive rate. Black line shows percent positive rate among the entire LG Health population as a comparison.
DISCUSSION
This study demonstrated that in a suburban hospital with a universally screened population, the percent positive for SARS-CoV-2 testing overall was 1.6%, with 68% of these cases being asymptomatic (approximately 1% of the total presenting population). Areas such as New York City that were epicenters early in the COVID-19 pandemic had reported rates of asymptomatic positive patients to be as high as 14% to 33% of the entire presenting population,4-6 leading to protocols that involved universal screening of obstetric populations due to the proclivity for viral spreading in this setting. However, in areas with much lower overall prevalence, the asymptomatic positive rate is expected to be much lower. This finding was confirmed in our study population.
For every symptomatic patient that tested positive during this time period, an additional two asymptomatic carriers of the virus were identified. This finding further highlights the difficulty in preventing the spread of SARS-CoV-2 in a younger adult population where infection with the virus is frequently asymptomatic.
BMI was the only demographic factor noted to be associated with a statistically significant increase in percent positive test results. Recent research suggests that the SARS-CoV-2 virus infects adipose tissue and there elicits an inflammatory response, which could lead to more severe disease in this population.12 This may partially explain the findings of our study. However, this finding should be interpreted carefully given the inaccuracy of BMI in pregnant individuals unless calculated prior to pregnancy.
The limitations of this study include low overall numbers of patients who tested positive, recall bias, inaccuracies in documentation of symptoms, and inability to distinguish between noncommunicable viral shedding with current testing protocols. The study also did not evaluate for symptom development following admission, but rather reflects patients who were asymptomatic at the time of testing. Additionally, the majority of the study period was conducted prior to the onset of widely available vaccines which may affect the rates of asymptomatic cases in the present day.
CONCLUSION
A universally screened population presenting for delivery in a suburban hospital demonstrated a 1.6% test positivity rate, with 68% of infections being asymptomatic. Approximately 1% of the overall presenting population was found to be an asymptomatic carrier. Although the prevalence of disease was lower in our study than in other populations, a large portion of the positive tests was asymptomatic. This finding adds weight to the importance of universal efforts to reduce viral transmission given the possibility of asymptomatic carriers to infect others.
ACKNOWLEDGEMENT
Thank you to the Lancaster General Health Research Institute, which provided support with study design, data collection, data analysis, data interpretation, and manuscript preparation.
REFERENCES
1. Palatnik A, McIntosh JJ. Protecting labor and delivery personnel from COVID-19 during the second stage of labor. Am J Perinatol. 2020;37(8):854-856.
2. Oran DP, Topol EJ. The proportion of SARS-CoV-2 infections that are asymptomatic. Ann Intern Med. 2021;174(9):1344-1345.
3. Johansson MA, Quandelacy TM, Kada S, et al. SARS-CoV-2 transmission from people without COVID-19 symptoms [published correction appears in JAMA Netw Open. 2021;4(2):e211383]. JAMA Netw Open. 2021;4(1):e2035057.
4. Bianco A, Buckley AB, Overbey J, et al. Testing of patients and support persons for coronavirus disease 2019 (COVID-19) infection before scheduled deliveries. Obstet Gynecol. 2020;136(2):283-287.
5. Breslin N, Baptiste C, Gyamfi-Bannerman C, et al. Coronavirus disease 2019 infection among asymptomatic and symptomatic pregnant women: two weeks of confirmed presentations to an affiliated pair of New York City hospitals. Am J Obstet Gynecol MFM. 2020;2(2):100118.
6. Sutton D, Fuchs K, D’Alton, M, Goffman D. Universal screening for SARS-CoV-2 in women admitted for delivery. N Engl J Med. 2020;382(22):2163-2164.
7. Campbell KH, Tornatore JM, Lawrence KE, et al. Prevalence of SARS-CoV-2 among patients admitted for childbirth in southern Connecticut. JAMA. 2020;323(24):2520-2522.
8. Goldfarb IT, Diouf K, Barth WH, et al. Universal SARS-CoV-2 testing on admission to the labor and delivery unit: low prevalence among asymptomatic obstetric patients. Infect Control Hosp Epidemiol. 2020;41(9):1095-1096.
9. COVID-19 by County. Centers for Disease Control and Prevention. Updated August 11, 2022. Accessed January 23, 2023. https://www.cdc.gov/coronavirus/2019-ncov/your-health/covid-by-county.html
10. Dong E, Du H, Gardner L. An interactive web-based dashboard to track COVID-19 in real time. Lancet Inf Dis. 2020;20(5):533-534.
11. Rakotosamimanana N, Randrianirina F, Randremanana R, et al. Gene-Xpert for the diagnosis of COVID-19 in LMICs. Lancet Glob Health. 2020;8(12):e1457-e1458.
12. Martinez-Colón GJ, Ratnasiri K, Chen H, et al. SARS-CoV-2 infects human adipose tissue and elicits an inflammatory response consistent with severe COVID-19. bioRxiv. Preprint posted online October 25, 2021.